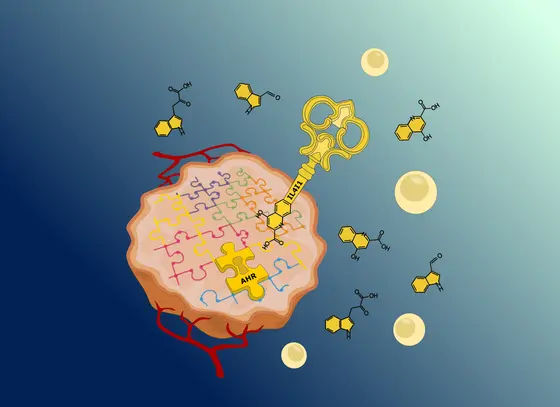
Immunotherapies activate the body's own defense against tumors and are currently revolutionising cancer therapy. Despite some groundbreaking successes, however, only a small number of patients benefit from the drugs currently available. The teams led by Christiane Opitz and Martina Seiffert, both at the DKFZ, and Saskia Trump from BIH, investigated the molecular mechanisms by which tumors escape destruction by the immune system. Their research results may provide important information for the development of new immunotherapy concepts.
The aryl hydrocarbon receptor (AHR) is also known as dioxin receptor because it mediates the toxic effect of dioxins. However, not only toxins, but also the body's own metabolic products can activate the receptor. An example of this are degradation products of the amino acid tryptophane, which we take in with our food as a building block of proteins. Tumors use these metabolites to their advantage: They promote the mobility of cancer cells and weaken the immune response against tumors.
However, the metabolic pathways by which the relevant tryptophane degradation products are produced have not been sufficiently researched. To find out more, the scientists systematically investigated in 32 different types of cancer which tryptophane-degrading enzymes are associated with an activation of the dioxin receptor.
One molecule in particular caught the scientists' eye: the enzyme IL4I1. No other enzyme of the tryptophan metabolism was as strongly linked to an activation of the dioxin receptor as IL4I1: “The metabolites formed by IL4I1 bind to the dioxin receptor and activate it, which leads to the suppression of immune cells,“ explains Saskia Trump, BIH.
Of clinical interest is the observation that the survival probability of patients with gliomas, a type of malignant brain tumor, decreased when the enzyme was present in higher concentrations in these tumors.
In a mouse model for chronic lymphatic leukaemia (CLL), a type of blood cancer, IL4I1 was shown to promote cancer through its effects on the immune system. “In animals that do not produce IL4I1 in the tumor environment due to genetic alterations, the immune system is significantly more successful in preventing the cancer from progressing,“ explains Martina Seiffert of DKFZ.
“IL4I1 has great potential as a target for drugs. So far, drugs that inhibit enzymes of the tryptophan metabolism have failed in clinical trials because the tumors did not respond to them. However, the role of IL4I1 has so far been disregarded and this enzyme has not yet been tested as a target molecule,“ said Christiane Opitz.
IL4I1 is a metabolic immune checkpoint that activates the AHR and promotes tumor progression. Ahmed Sadik*, Luis F. Somarribas Patterson*, Selcen Öztürk*, Soumya R. Mohapatra*, Verena Panitz, Philipp F. Secker, Pauline Pfänder, Stefanie Loth, Heba Salem, Mirja Tamara Prentzell, Bianca Berdel, Murat Iskar, Erik Faessler, Friederike Reuter, Isabelle Kirst, Verena Kalter, Kathrin I. Foerster, Evelyn Jäger, Carina Ramallo Guevara, Mansour Sobeh, Thomas Hielscher, Gernot Poschet, Annekathrin Reinhardt, Jessica C. Hassel, Marc Zapatka, Udo Hahn, Andreas von Deimling, Carsten Hopf, Rita Schlichting, Beate I. Escher, Jürgen Burhenne, Walter E. Haefeli, Naveed Ishaque, Alexander Böhme, Sascha Schäuble, Kathrin Thedieck, Saskia Trump#, Martina Seiffert#, Christiane A. Opitz#. Cell 2020,
DOI: doi.org/10.1016/j.cell.2020.07.038