Until now, scientists have failed to establish a link between the properties of pancreatic cancer, such as its aggressiveness, and changes, i.e. mutations, in the tumor's genome. Moreover, pancreatic cancer forms metastases much faster than other types of cancer. Here too, the genetic causes are unclear.
A team headed by Professor Dieter Saur of the German Cancer Consortium (DKTK) and of Professor Roland Rad of TUM University Hospital rechts der Isar and DKTK has taken an important step towards solving both mysteries. With the help of various mouse models for pancreatic cancer, they have succeeded in elucidating the molecular pathways of tumor development in detail and have gained a better understanding of how various characteristics of the disease arise. The study was published in the journal Nature.
Tumor cells have multiple defective copies of a cancer gene
Healthy cells in humans possess two copies of each gene. For their experiments, the researchers mutated one of the two copies of the KRAS gene in mice. The gene plays a key role in cellular proliferation and is activated in 90% of all human pancreatic tumors. Such genes are referred to as oncogenes. The team headed by Roland Rad made a surprising discovery: The mutant gene was often duplicated even in very early stages of the cancer. In cases where a tumor had not doubled the mutated KRAS gene copy, the researchers discovered duplications in other cancer genes.
“It therefore appears that the cell amplifies the growth signal due to the presence of extra gene copies. This model of dosage amplification during tumor development had not previously been considered,“ says Sebastian Müller, lead author of the study. “We also showed that as the number of mutant KRAS copies increases, the tumor's aggressiveness and ability to metastasize also increases.“
Disruption of endogenous protective mechanisms determines the evolution of the cancer
Normally, healthy cells have their own protective mechanisms to prevent mutations from accumulating. So how could the cells develop such dosage amplification without being prevented from doing so?
“This shows the importance of mouse models, which allow us to closely observe and experimentally review the extraordinarily complex processes of cancer development at the molecular level: from healthy cells to cancer precursors through to aggressive tumors and their spread to other organs,“ Professor Dieter Saur explains.
After the KRAS mutation was induced by the researchers, other mutations in what are known as tumor suppressor genes developed. A healthy cell possesses a whole series of such protective genes to prevent cancer from developing. A significant finding by the team was that either the mutant KRAS gene or another cancer gene is amplified, depending on which tumor suppressor gene is affected and to what degree its function is impaired.
Key developmental stages explained
Only after the cell's inbuilt protective mechanisms have been switched off and dosage amplification occurs does a tumor ultimately form. Which pathway the cell follows, and which genes are involved then largely determine the characteristics of a pancreatic tumor.
For the first time, the dosage amplification model allows us to identify genetic patterns that explain a tumor's aggressiveness and metastasis. “We have indications that our discovery constitutes a fundamental principle in the development of tumors and plays an essential role in other cancers. We're now investigating the extent to which these new insights into cancer biology can be used to develop new therapeutic strategies,“ says Professor Roland Rad, explaining the team's next research goals.
---
The following institutions contributed to the study: Technical University of Munich (Central Institute of Translational Cancer Research, Clinic and Policlinic for Internal Medicine II, Institute of Pathology), DKTK and DKZF Heidelberg; The Wellcome Trust Sanger Institute, Cambridge; Ludwig-Maximilians Universität (Anthropology & Human Genomics und Innere Medizin II des Klinikums Großhadern), Helmholtz Zentrum München (Research Unit Radiation Cytogenetics); Universidad de Oviedo (Bioquímica y Biología Molecular, IUOPA und CINN-CSIC), Instituto de Medicina Oncológica y Molecular de Asturias (IMOMA), Oviedo, University of Cambridge (Department of Veterinary Medicine), Instituto de Medicina Oncológica y Molecular de Asturias, Instituto de Biomedicina y Biotecnología de Cantabria.
Original publication
S. Mueller, T. Engleitner, R. Maresch, M. Zukowska, S. Lange, T. Kaltenbacher, B. Konukiewitz, R. Öllinger, M. Zwiebel, A. Strong, H.-Y. Yen, R. Banerjee, S. Louzada, B. Fu, B. Seidler, J. Götzfried, K. Schuck, Z. Hassan, A. Arbeiter, N. Schönhuber, S. Klein, C. Veltkamp, M. Friedrich, L. Rad, M. Barenboim, C. Ziegenhain, J. Hess, O. M. Dovey, S. Eser, S. Parekh, F. Constantino-Casas, J. de la Rosa, M. I. Sierra, M. Fraga, J. Mayerle, G. Klöppel, J. Cadiñanos, P. Liu, G. Vassiliou, W. Weichert, K. Steiger, W. Enard, R. M. Schmid, F. Yang, K. Unger, G. Schneider, I. Varela, A. Bradley, D. Saur, R. Rad, Evolutionary routes and KRAS dosage define pancreatic cancer phenotypes, Nature, 2017, DOI: 10.1038/nature25459
www.nature.com/articles/nature25459
A picture is available for download
mPDAC.jpg
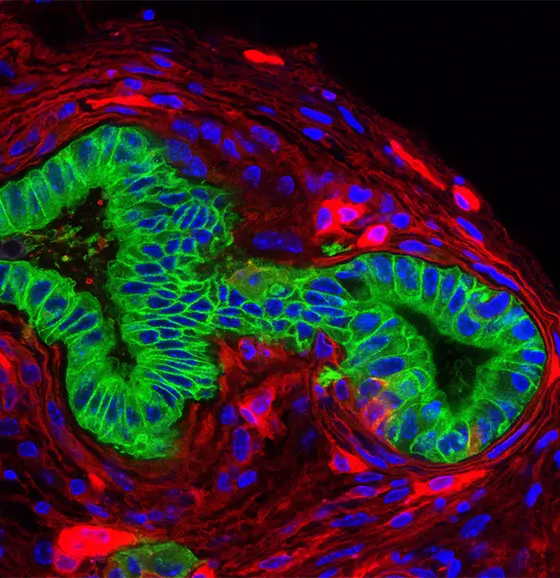
Caption: Pancreatic Carcinoma (green) in a mouse line with mutated Kras gene. Cells of tumor microenvironment are shown in red.
Note on use of images related to press releases
Use is free of charge. The German Cancer Research Center (Deutsches Krebsforschungszentrum, DKFZ) permits one-time use in the context of reporting about the topic covered in the press release. Images have to be cited as follows: “Source: Dieter Saur/ TUM“.
Distribution of images to third parties is not permitted unless prior consent has been obtained from DKFZ's Press Office (phone: ++49-(0)6221 42 2854, E-mail: presse@dkfz.de). Any commercial use is prohibited.