Research Group: Neuro-oncologic Imaging (Radbruch)
Research Focus:
The research group Neuro-oncologic Imaging focuses on the one hand on the application of new MRI sequences in patients with neuro-oncologic diseases using PET-MR, 3 and 7 Tesla MRI and on the other hand on the application of advanced computational methods such as radiomics and computer assisted diagnosis.[1-4] The research group has a strong emphasis on translational research and regards itself as a connecting link between MRI physics, computer science and clinical application.
The goal of our work is to improve the characterization of tumor-growth and tumor-responses to different therapies in order to establish response criteria for daily clinical decision making. This is not only of major importance for individual treatment decisions but also for the correct assessment of clinical studies that increasingly rely on the correct assessment of imaging parameters. Our research group uses the complete spectrum of MRI technology such as Diffusion-, Perfusion-, Susceptibility-weighted- MRI, spectroscopy and Chemical Exchange Saturation Transfer MRI. This multi-modal approach is combined with advanced post-processing methods and consequently used on clinical questions.
Research Project: Characterization of Tumor Vessels Using TOF-MRI at 7 Tesla (Paech/Radbruch)
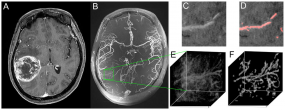
Figure 1. Demonstration of multiscale vessel enhancement filtering. A) Contrast enhanced T1 weighted images at 3 Tesla of a patient with right temporal glioblastoma. B) 7 Tesla ToF angiography maximum intensity projection. Within the selected VOI (E) (representative slide in (C)), vessels are automatically detected and visualized (F) (representative slide with red colored vessels in (D)).
© dkfz.de
Generally, high grade glioma are characterized by vascular proliferation of tumor vessels that potentially differentiate them from other brain tumors such as low grade glioma. Hence, direct visualization of tumor vessels could potentially contribute to the differential diagnosis of glioblastoma. Beyond this, a quantification of the tumor vasculature could be used for the assessment of antiangiogenic therapies, since an initial normalization of tumor vasculature during therapy response has been reported.
The differentiation of patients who benefit from anti-angiogenic treatment and patients who only suffer from side effects currently remains an unsolved problem within daily clinical decision making. Hence, imaging biomarkers that stratify responders from non-responders are highly needed and TOF-Angiography at 7 Tesla might contribute to enable this stratification.
Research Project: Automatic brain metastases detection in patients with malignant melanoma on contrast-enhanced T1w MRI using deep neural networks (Deike-Hofmann/Radbruch)
Brain metastases are the most frequent brain tumors, most often caused by breast cancer, lung cancer and by malignant melanoma. Previous studies demonstrated the suitability of using spherical templates to detect brain metastatic lesions, which are often observed as bright spherical-like solid masses on contrast-enhanced MRI. However, a common difficulty that most of the studies that used contrast-enhanced imaging faced was hyperintensities found in blood. Signals from blood frequently generate false positives and decrease the diagnostic accuracy of computer assisted diagnosis systems in detecting lesions in the brain.
Subtraction post-processing improves the detection of signal intensity. Generally, Software can be used to automatically subtract pixel values of a non-enhanced dataset from those of a contrast-enhanced dataset to produce an image that emphasizes areas of contrast enhancement, e.g. intracerebral vessels as well as disruption of blood-brain barrier in brain tumors. Therefore, the segmentation of blood vessels is facilitated in subtraction series as well as detection of contrast-enhancing lesions.
The current study investigates whether the imaging features of contrast-enhanced MRI after subtraction post-processing and vessel segmentation can be applied to reduce false positives from blood and increase the sensitivity for small brain metastases with computer assisted diagnostic approaches.
Literature:
1. Radbruch A, E.O., Wiestler B, Paech D, Burth S, Kickingereder P, Nowosielski M, Bäumer P, and S.H. Wick W, Bendszus M, Ladd M, Nagel AM, Heiland S, Quantification of Tumor Vessels in Glioblastoma Patients Using Time-of-Flight Angiography at 7 Tesla: A Feasibility Study. Plos One, 2015.
2. Paech, D., et al., Nuclear overhauser enhancement mediated chemical exchange saturation transfer imaging at 7 Tesla in glioblastoma patients. PLoS One, 2014. 9(8): p. e104181.
3. Zaiss, M., et al., Relaxation-compensated CEST-MRI of the human brain at 7T: Unbiased insight into NOE and amide signal changes in human glioblastoma. Neuroimage, 2015. 112: p. 180-8.
4. Paech, D., et al., T1rho-weighted Dynamic Glucose-enhanced MR Imaging in the Human Brain. Radiology, 2017. 285(3): p. 914-922.
